 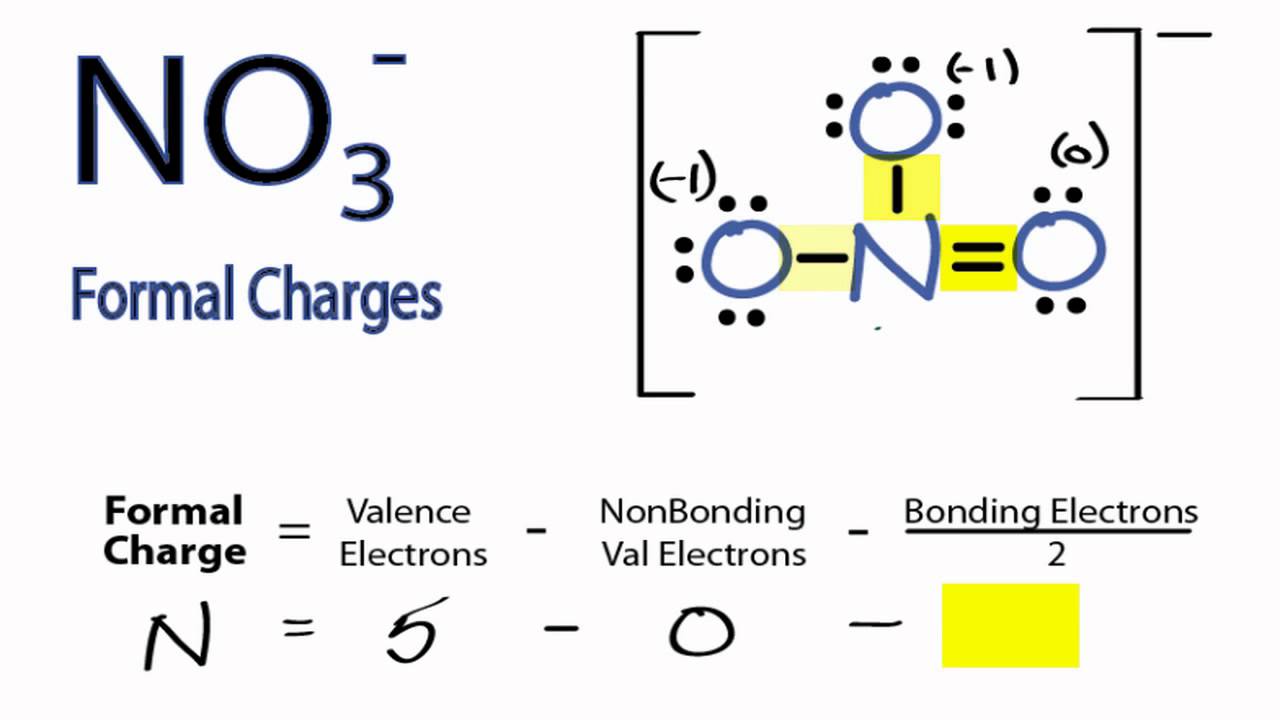
In reality, the distribution of electrons in the molecule lies somewhere between these two extremes. 
Oxidation states overemphasize the ionic nature of the bonding the difference in electronegativity between carbon and oxygen is insufficient to regard the bonds as being ionic in nature. The oxidation state view of the CO 2 molecule is shown below: With the oxidation state formalism, the electrons in the bonds are "awarded" to the atom with the greater electronegativity. This can be most effectively visualized in an electrostatic potential map. The covalent (sharing) aspect of the bonding is overemphasized in the use of formal charges, since in reality there is a higher electron density around the oxygen atoms due to their higher electronegativity compared to the carbon atom. The formal charge view of the CO 2 molecule is essentially shown below: With formal charge, the electrons in each covalent bond are assumed to be split exactly evenly between the two atoms in the bond (hence the dividing by two in the method described above). The reason for the difference between these values is that formal charges and oxidation states represent fundamentally different ways of looking at the distribution of electrons amongst the atoms in the molecule. If the formal charges and oxidation states of the atoms in carbon dioxide are compared, the following values are arrived at: The concept of oxidation states constitutes a competing method to assess the distribution of electrons in molecules. 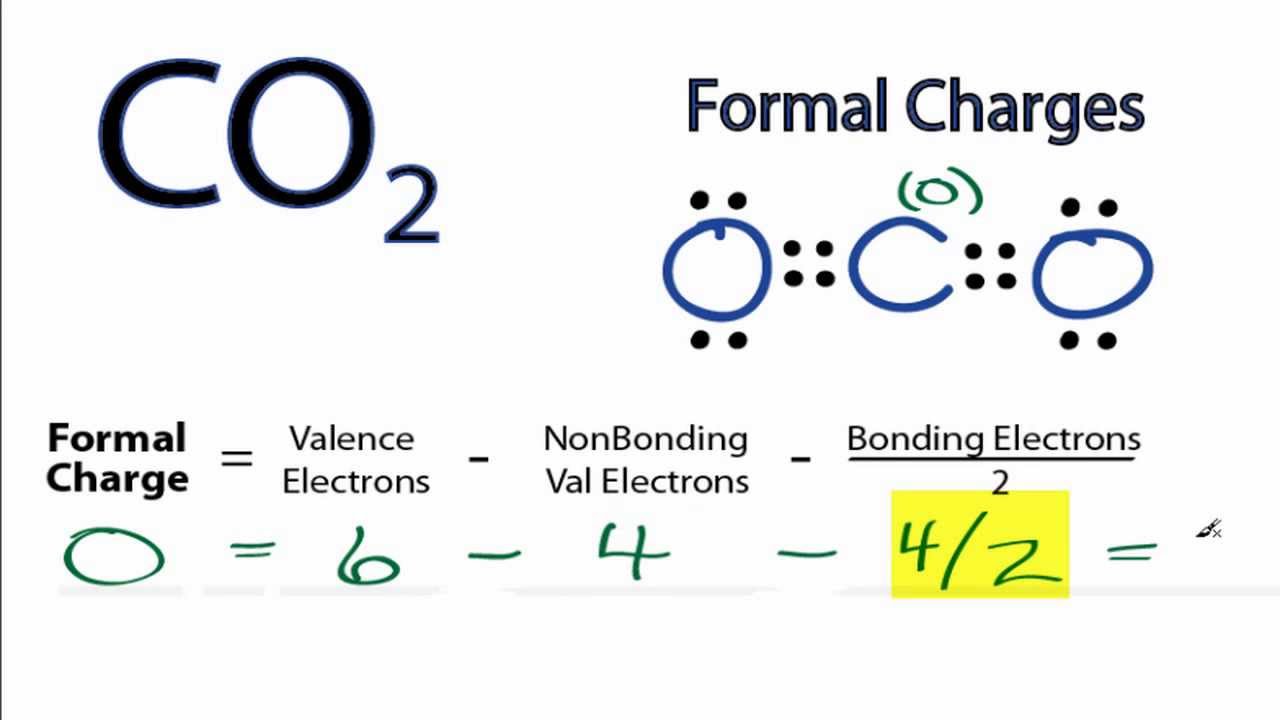
\)įormal charge compared to oxidation stateįormal charge is a tool for estimating the distribution of electric charge within a molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
